About This Episode
Can AI help us model biology down to the molecular level? Neil deGrasse Tyson, Chuck Nice, and Gary O’Reilly learn about Nobel-prize-winning Alphafold, the protein folding problem, and how solving it could end disease with AI researcher and Chief AI Officer of Isomorphic Labs, Max Jaderberg.
We break down the protein folding problem and why it was so hard to crack. How do we learn to decode how strings of amino acids fold into complex, dynamic shapes that power our biology? How did AlphaFold crack this decades-old puzzle? We explore how Alphafold 3 uses the shapes of proteins to predict protein-drug interactions. How close are we to using AI and deep learning to design new medicines instead of discovering them by chance? Could generative AI invent molecules that precisely target the cause of a disease?
We take these ideas into the future: Could AI help us fix misfolded proteins that cause illness? Can we use these tools to create personalized drugs tailored to your unique genome? Will it make it economically feasible to treat rare diseases? Could this even help us slow aging or regenerate limbs, like salamanders do? Could this technology combat cancer, fix rare genetic disorders, or even digest plastic? And with quantum computing on the horizon, what happens when we combine it with AI? Will chemistry ever be done the same way again?
Thanks to our Patrons Riley r, pesketti, Lindsay Vanlerberg, Andreas, Silvia Valentine, Brazen Rigsby, Marc, Lyda Swanston, Kevin Henry, Roberto Reyes, Cadexn, Cassandra Shanklin, Stan Adamson, Will Slade, Zach VanderGraaff, Tom Spalango, Laticia Edmonds, jason scott, Jigar Gada, Robert Jensen, Matt D., TOL, Thomas McDaniel, Sr., Ryan Ramsey, truthmind, Aaron TInker, George Assaf, Dante Ruzinok, Jonathan Ford, Just Ernst, David Eli Janes, Tamil, Sarah, Earnest Lee, Craig Hanson, Rob, Be Love, Brandon Wilson, TJ Kellysawyer, Bodhi Animations, Dave P., Christina Williams, Ivaylo Vartigorov, Roy Mitsuoka (@surflightroy), John Brendel, Moises Zorrilla, deborah shaw, Jim Muoio, Tahj Ward, Phil, Alex, Brian D. Smith, Nate Barmore, John J Lopez, Raphael Velazquez Cruz, Catboi Air, Jelly Mint, Audie Cruz for supporting us this week.
NOTE: StarTalk+ Patrons can listen to this entire episode commercial-free.
Transcript
DOWNLOAD SRTSo AI was not satisfied just whooping our ass in chess and in Jeopardy and everything else where it looks like brains mattered.
It’s now taken over our physiology.
Well, no, you pointed it in a good direction.
Aimed it at a good place, and we’re getting somewhere.
To solve our diseases.
Yeah, so now it’s going to cure us of all disease before it makes us its slaves.
Because we need a healthy slave.
All that and more coming up on StarTalk.
Welcome to StarTalk.
Your place in the universe where science and pop culture collide.
StarTalk begins right now.
This is StarTalk Special Edition.
Neil deGrasse Tyson, your personal astrophysicist.
Special Edition means we’ve got Gary O’Reilly in the house, Gary.
Hi, Neil.
Former soccer pro.
Apparently.
Yeah, and soccer announcer.
Yes, definitely.
And you still do that, don’t you?
I do.
Chuck Nice, baby.
Hey, announcing that I know nothing about soccer.
You’re in my club then.
Announcing that you are American.
American dog on a drill football.
Violence.
So we’re talking about AI today.
Yeah.
That’s a favorite topic.
We revisit that often.
Only the future of the entire world.
AI as it matters in biology.
Oh, wow.
Now that’s a big deal.
I know.
I know.
Because people are thinking about composing your term paper or winning a chess.
But it’s got a whole frontier ready to be explored.
And so tell me what you and your producers cooked up today.
Okay, so we’ve been on the case to get these guys involved for some time, but they are so busy.
So here we go.
I’ll say it.
I’m made of proteins.
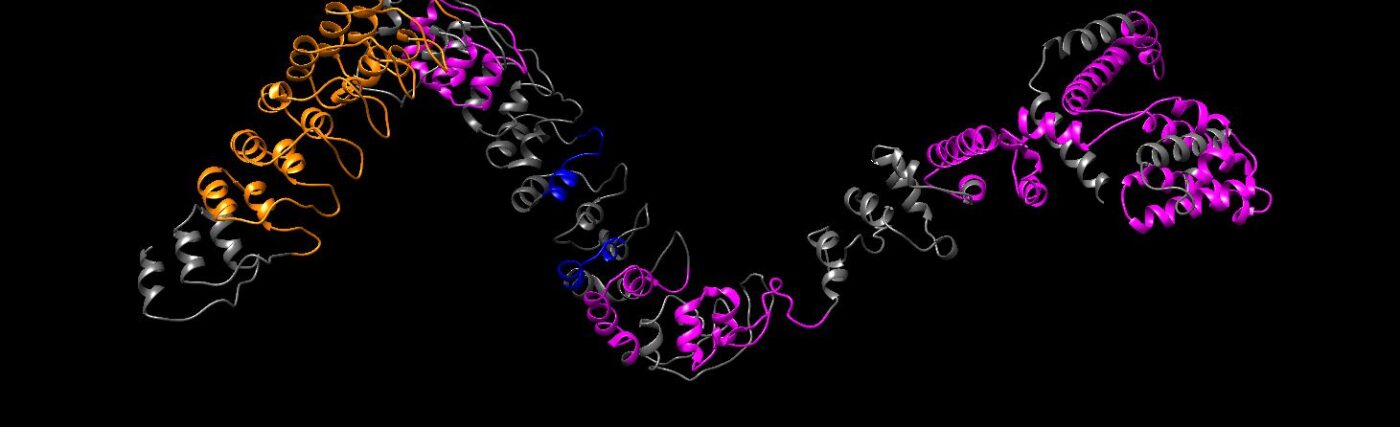
Yes, you’re made of proteins from strings of amino acids that fold into shapes that put all together form us.
But there’s a fundamental problem in biology that has implications for all of medicine.
How do these proteins fold up?
For this solution, we looked at AI and a Google DeepMind talk called AlphaFold.
The second iteration of AlphaFold 2 won the Nobel Prize in Chemistry last year for answering this very question.
Who knew AI was smart?
We should say AI won all the Nobel Prizes.
Yeah, just give them to them now, just park them all up.
Now the isomorphic labs together with Google DeepMind developed and released AlphaFold 3, yes, we’re on the third iteration.
That was last year and applied these new AI models for drug discovery.
Oh, that’s great.
All right, so think this through.
Could our next generations of treatments be computer generated?
Oh, yeah.
Oh, by the way, Neil, let’s introduce our guest.
I will.
We’ve got Max Jaderberg.
Did I pronounce that correctly?
Yeah, you got it.
You got it right.
Let’s hear you say it.
Let’s hear you say it.
Me say it.
Max Jaderberg.
Oh, he got it perfect.
That’s what I got.
He copied you.
He was practicing.
He was practicing.
So you studied AI at Oxford?
That’s right.
That’s right.
Now I hear that’s a community college.
Oxford Community College.
That’s exactly right, in Oxford, England.
Yes.
So specialized in deep learning algorithms, I got your little bio here, for understanding images.
Nice.
That was a big advance when a search can go into an image.
I thought I’d died and gone to computer heaven when that started.
Yeah, yeah.
I mean, this was 10, 15 years ago, back before AI was cool.
Right, right.
Where, you know, you talk about AI and it’s something from a sci-fi book, but understanding images and videos was like the big thing at that point in time.
We couldn’t actually do that very well.
I searched my 9,000 images on my computer for the word telescope.
Yeah.
And it found telescope written in Chinese.
That’s crazy.
On a photo taken at an angle in one of my images.
When I was visiting China.
Yeah, this is it.
Like during my PhD, we took all of the BBC’s back catalog.
No.
And we ran my algorithm across it and created a search engine.
So you could pull up footage from decades ago that had this text or these objects.
That’s seriously some archive.
If you went through the BBC.
When you do that, do you tie?
So when the AI is looking at an image, it’s not seeing the image the way we do.
We’re not even seeing a whole image.
Our brains, we’re really just intuiting an image when we see it as our brain.
That’s how we do it.
But the AI actually…
It’s like a holistic processing.
It’s a holistic processing.
AI actually sees the image and what it’s seeing is pixels.
That’s right.
And really all it’s doing is just, oh, this pixel, this pixel, this pixel, in this arrangement, that’s this image.
So do you tie that to language and that’s how we search?
Or is the search just the AI knows the actual image itself?
This was like the big breakthrough back then, and I was doing my PhD and this is what deep learning as well is all about.
Right.
You can imagine if you have this pixel, this image full of pixels, how do you actually code up how to read text from there?
How do you tie it to the language of the text?
Right.
It’s unimaginably hard to code that up by hand.
Yes.
So instead, what you do is you put these, what they’re called neural networks, they look at all of the pixels of the image, and you give it lots and lots of examples of images that have some way it’s got the text in it, and you tell the neural network what the text is.
I see.
And the neural network, through lots and lots of training, starts to work out its internal algorithm to extract the information from these pixels, piece it all together, and spit out the actual text, or spit out what the objects are.
Wow.
So you’re currently chief AI officer at Isomorphic Labs.
This is a biology place.
That’s right.
Do you have any biology in your background?
Formal biology?
No.
No, okay.
So they want you for your AI.
That’s right, that’s right.
So I was at a place called DeepMind beforehand.
Google.
Google DeepMind, exactly.
I was there for a long time.
I absolutely love this core AI technology called deep learning.
That’s what I’ve been developing my whole career so far.
At DeepMind, we were working on some crazy stuff, learning to play chess and go and beating top professionals at games like StarCraft.
Back then, it was about…
All of it was a big deal.
Yeah, and because the world didn’t know what AI was, so we were trying to prove that this was even a thing.
Right?
It seems crazy now, but back then, it was just proving that this was actually a real thing.
But at the core, I love this technology.
I want to see it have profound impact on our world.
And I was thinking these things…
That’s where it begins.
Yep.
That’s where the terminator starts.
It’s always the innocent dreamer who says, this can change the world for such good.
And it’s in my closet now.
Would you like to see?
Exactly.
And then it’s always like some evil businessman who’s just like, with my weather machine, I will one day rule the world.
Apart from that, carry on.
The good thing is, there’s some pretty strictly good applications of AI that we can drive.
Demis Asabis started Isomorphic Labs, spinning that out of Google DeepMind, to really think how can we apply AI to actually completely solve all disease.
Okay, so it has genetic links back to its origin story.
Exactly.
It was actually DeepMind.
I feel better about that now.
Yeah.
Okay.
You happy?
I’m happier now, yes.
So I moved over as part of that founding team to head up AI in this space, and it’s been about three and a half years now.
Been a crazy journey, but it’s fascinating.
It’s so much fun.
So you got this AI expertise, and Alpha Fold spins off this biological application of it.
First, tell me the word isomorphic.
What does that mean in biology?
Isomorphic is this technical term which is a one-to-one mapping of space, right?
And the reason we’re called isomorphic labs is really that we believe that biology is really, really complicated.
In the world of physics, we can write down equations for physics with maths.
And maths is that perfect description language for physics, but you can’t really just write down equations in maths for biology, for the cell.
It’s just too complicated.
Biology is the most complex expression of chemistry that we know.
There’s just so many moving parts.
Did you just make that up?
So we’re looking for a Rosetta Stone here for the language of biology.
Exactly.
So what could be that perfect description language for biology?
We believe AI and machine learning is that.
So there could exist an isomorphism, a mapping between the biological world and the world of AI machine learning.
Hence the name.
Gotcha.
All right.
So tell us about protein folding.
Because when we learn about chemistry, we learn about chemical reactions.
And we’re not really taught that the shape of the molecule should have anything to do with anything.
It’s just what is the chemical symbol.
And when you write down the chemical equations, there’s no shape in there.
There’s just what elements and molecules comprise it.
And those equations don’t really ever represent the three-dimensional nature.
Exactly.
You don’t even know if it has handedness, right?
So take us from there.
We think about proteins.
Proteins are these fundamental building blocks of life.
They’re inside of everyone.
They make up everything we have basically.
And they’re made up of what’s called a sequence of amino acids.
Each amino acid is a molecule.
There’s about 20 different amino acids.
And you put them together in a long…
Ever or just in life?
In life, you can have non-natural amino acids as well that you can make as well.
You can make them?
You can make them.
And actually use those for drugs sometimes.
You string these amino acids together, and that becomes a protein.
But they don’t exist as these strings.
They fold up spontaneously in the cell to create these 3D shapes.
And why that’s important is that these proteins, they’re basically molecular machines.
They don’t just exist by themselves.
They actually create these little pieces of machinery.
They interact with other proteins.
They interact with other biomolecules like DNA and RNA.
That interaction is a shape fitting.
Exactly, exactly.
It’s a puzzle.
It’s a 3D puzzle.
It’s a 3D puzzle.
Exactly.
It’s a 3D jigsaw puzzle.
And it’s not static.
Which is way harder than a 2D jigsaw puzzle.
And these are not static things.
It’s not just static puzzle pieces coming together.
They change shapes.
Something comes in contact, and that opens up something else on the other side of the protein, which changes the machine, and on and on it goes.
And that’s why I was going to ask you, what speeds is this folding taking place?
Is it continuous?
Once it folds, that’s it?
But you’ve just told me, no, it just keeps moving through the whole thing.
Yeah.
These are really, really complex, dynamical systems, you know, composed of thousands, millions, trillions of atoms within our cells, unfolding over the course of, you know, microseconds and beyond.
And this dino-cism that you’re talking about does…
What word is that?
Dino-cism?
That’s a word?
Isn’t that a word?
Dino-cism?
I think I just made it up.
Mism.
Oh, dino-mism.
No, dynamism.
Dynamism.
Thank you.
Thank you.
I’m correcting grammar.
This is a first.
Dynamism.
Dynamism.
It’s a different kind of dinosaur.
Dynamism.
Not dinosaur.
Dinosaur.
Not dinosaur.
Not dinosaur.
But the dynamism that you’re talking about within the cell, when you look at each one of us, since each one of us is so different, even though there’s a general, like, execution and blueprint, we all come out so different.
Is that part of the process that you are looking at and mapping?
I would say to a jellyfish, we all look identical.
True.
Okay?
They’re not saying, oh, is your skin color slightly different or you’re slightly taller.
Yes.
You’re describing functions at a cellular level.
Is your job to understand that or is your job to figure out extra ways to fold proteins that maybe biology has yet to even figure out, that can then solve problems that we encounter that the natural universe has not.
So, that was a good question.
I’m so happy with yourself.
I’m happy today.
I’m so happy with myself today.
Yeah.
This is really interesting.
We have these little molecular machines, these proteins, and we care about that 3D structure and how they work for two reasons.
One, we want to understand how our cells work, because if something goes wrong with that, which is the case for disease, then we want to understand, okay, where can we, where do we actually need to go in and start fixing that?
Right.
Or how we can stop it from actually going wrong in the first place.
Exactly.
Exactly.
So, that’s one thing.
And then when we think about, okay, how can we go and fix that?
What we’re actually saying when we’re doing drug design, we’re saying, can we create another molecule that will come into the cell and actually start modulating these molecular machines?
This drug molecule is going to actually attach to this protein over here, and that’s going to cause this protein to change shape, for example, and so it won’t operate how it normally does.
Or so we stop that protein working.
Or we make it work better.
These are the sort of things we do in drugs.
That kind of reminds me of messenger RNA vaccines that we developed for COVID.
Yeah.
You know, there’s so many different types of molecular mechanisms that we take advantage for for drug design.
Are the folding proteins generally following a set pattern in the way that they do fold?
And you’re able to map them, and when they misfold, that’s when you’re able to flag that up, or have I just reinvented something or talked rubbish?
That would be cool.
No, no, you’re on to something.
That would be great.
So, you know, like, the amazing thing is that we can actually, turns out, predict how these proteins fold.
So they are…
So you’re modeling that process.
We’re modeling that with deep learning, with neural networks.
That’s what alpha fold, you know, and all its generations are all about.
And that means that we can actually just take in a sequence of amino acids, knowing nothing about this protein before, and then get out the 3D structure.
And normally this would take people months, if not years, to work out this 3D structure.
So how is it that alpha fold knows how a large molecule wants to fold?
Again, it’s gotta know that in some way.
It’s learned this from a few hundred thousand examples.
So chemists, biochemists over the last 50 years, they’ve been working out these protein structures by hand.
They’ve been literally synthesizing protein, crystallizing it, then shooting x-rays at this to like look at the electron scattering.
And from that, you can resolve the protein structure.
It’s a pretty hard process.
But people have been doing that.
That’s your way to photograph what the shape of the molecule is.
That’s your way to photograph it in reality.
With that kind of, it’s basically an electron microscope at that level.
Yeah, similar.
It’s like electron scattering, yeah, exactly.
And so people have been doing that for the last 50 years and depositing these structures.
And now we’ve taken all of that data and trained a neural network to go just from the input of what is this molecule description to try and predict all of that data.
And the amazing thing is, and this is really remarkable, is that you can then train this on the last 50 years of data.
That’s a couple of hundred thousand protein and biomolecular systems.
But you can apply it seemingly to everything we know about in the protein universe, in the proteome.
Well, it has the…
The proteome.
Oh, we like that.
The proteome.
Yes, the proteome.
So how accurate is AlphaFold, and AlphaFold went on the third iteration with its predictions?
Because AI has been around a little while, as you’ve already said, and you’re not the only AI tool that’s out there.
But how accurate is this particular tool?
Yes.
So AlphaFold 2 was that big jump where we started to get experimental level accuracy for just proteins.
And that’s what won the Nobel Prize in Chemistry.
So you balanced it off against empirical experimental…
The benchmark is doing the real lab work itself.
So AlphaFold 2 reached that level.
Now AlphaFold 3 expands from just proteins to incorporate other biomolecular types.
So proteins with other proteins, proteins with DNA, with RNA, with what’s called small molecules, which are…
They start mixing all that up.
Or maybe not the neighborhood.
Maybe that neighborhood gets a little bit of an upgrade.
No, that’s when you make the superhuman.
So everybody thinks it’s going to be the Terminator.
It’s not going to be the Terminator.
It’s going to be the superhuman.
And then they’re going to be like us.
And they’re going to look down on us and go, you know, why do we need you guys?
And that’s it.
So anyway, you’re able to predict these.
And have you actually taken any of the modeled predictions and made the proteins?
Yeah.
Oh, yeah.
Or tell us where you expect these to lead to new and innovative drugs.
Because otherwise, it’s just a puzzle exercise.
Yeah.
It’s a great Lego set.
Lego set.
We want the guests to enjoy this.
It’s like, oh, my God, how much that Lego set costs?
Only 10 billion dollars.
Sorry, go ahead.
Yeah.
So if you take a particular disease, and we think that we can actually solve this disease by modulating a particular protein, the question is how we do that.
We design a drug molecule and we want it to fit to this protein in a certain way.
So this is where traditionally you would have to actually either just guess, or go into the lab and crystallize each one of those combinations, and then photograph them and see if it worked.
But now you can model it and the AI can do a thousand of those in like a minute.
Isn’t this what you call the target proteins?
Yes.
Wow.
So if you know you’ve got a certain target protein, do you not then run that against a list of drugs and think, this one, drug A works better with this, or maybe it’s drug D or whichever letter of the alphabet you’re on.
And now we’ve become the sort of detective.
And has this Alpha Fold 3 produced how many clues and how many answers, or are we still grappling with?
That’s interesting.
Yeah.
Instead of trying to figure out the drug, the AI actually figures out the drug for you.
Drug discovery.
Well, exactly.
You know, if you let the guy speak, we might hear the two of you.
Okay, cool.
We’re figuring out this whole industry ourselves, Gary.
Yes.
Yeah, you know, this is exactly where it’s going.
So we can start actually rationally designing these drugs.
Traditionally, you would take a million random molecules and you would just throw them at these proteins and see what sticks.
And that’s how so many drugs have been created historically.
You go back further and you’re sifting through mud to find these sort of molecules.
This is why there’s been such a low percentage of success rates with the sort of drugs for whatever the problem is.
That’s part of it because we don’t necessarily understand how these molecules are working.
But with something like Alpha-Fold 3, you can put the molecule, put the target protein into the system, into the neural network, and you get out the 3D structure.
And as a chemist, you can start to understand, okay, how is this small molecule drug modulating this protein?
Now, still the problem is, well, how do you find that small molecule in the first place that’s going to be good for this protein?
It’s estimated there’s like 10 to the power of 60 possible drug-like molecules out there.
That’s 10 with 60 zeros.
So even if-
People know what 10 of the 60 is.
Yeah, yeah, yeah.
So-
Don’t be rude.
Even if you had the perfect Alpha-Fold, you’d have to run that across 10 to the power of 60 molecules, which is just computationally impossible, it’s unfeasible.
Until quantum computing.
And so then what we need is something that we call a generative model or an agent, which is able to actually search through that space, understand that entire molecular space and come up with molecule designs for you.
Oh, cause the 10 of the 60 is if you just did it randomly.
Exactly.
Right, right.
That’s just drawn in at anything.
Right, right.
If you don’t do it randomly, then you can-
Yeah, but randomly is the state-of-the-art method.
That’s how people do it.
I mean, it’s how people currently do it.
It’s how people currently do it.
Well, he called it state-of-the-art.
You’re the state-of-the-art.
Right, thank you.
Let’s use the word properly here.
So what if the protein turns left when you’ve mapped it to turn right?
Is that when we have issues that even Alpha Phi 3 has a problem with?
Exactly.
These are not perfect models at the end of the day.
They’re very, very accurate, but they will make some mistakes.
So you still do, currently, need to go into the lab occasionally, but the amount of lab work you have to do is so much less.
Right.
And often you can find the area of molecular space where these models work really, really well.
And we then go out to the lab later down the line.
We crystallize these things and we see, yeah, like this is a perfect mapping of what the model predicted.
So back to an earlier point, in the old days, like last month, you, the pharmaceutical companies, Big Pharma, would spend millions, maybe not quite a billion, hundreds of millions of dollars developing a drug.
We think that holding aside what might be abuses of pricing, the fact that there’s some truth to this first pill cost 50 million dollars.
The second pill cost 10 cents, because they had to research to get the formula for that first pill.
If you have narrowed the search space, then the cost of developing that first pill can be manifold smaller.
It costs, on average, three billion dollars to create a new drug.
Wow.
That’s on average.
I said, yeah, there’s a-
So I was low when I said $100 million.
You were low-balling.
I was low-balling it, okay.
So this is a massive opportunity to like completely change just like the cost, the speeds, the business model, and the business model as we do that.
Is it proprietary?
So here’s my real, because here’s where you would revolutionize.
So if I come up with it and I’m company A, right?
It’s mine and I get to determine everything.
If you’re an AI company and you’re just doing this, okay, so that you can sell it, then it’s yours.
Which one will make prices lower for the consumer?
Our goal is to really redefine this way you do drug design.
So it becomes so much cheaper.
We have so much more abundance of potential drugs and chemical matter that it really does change the business model.
It changes the economics of the space.
So you can actually revolutionize the cost of making drugs.
Yeah, that’s where we’re going.
Okay, all right.
Is one of the next steps with AlphaFold, whichever it’s three or maybe the next iteration or so, going to investigate why and what drives the misfolding of a protein so as you can kind of get ahead of even the story of that happening?
Wow.
So actually, the misfolding of a protein is another thing.
That’s what causes some types of disease where you’ll have a genetic mutation, a mutation in your DNA which will change a particular amino acid in that protein, and so it doesn’t fold the normal way it should fold, and so it doesn’t function as it normally should as a molecular machine.
And so things like alpha fold can help us understand what are those mutations that cause misfolding, they’re called missense mutations, and then these could be potential drug targets, so we could think about molecules that could mitigate against that.
If I understand correctly, if you look at the PDR, it is this thick, and the physician’s desk reference is this thick, so it’s the size of an old-style Manhattan phone book, okay, it’s very thick, multiple inches across, and it’s chock full of existing medicines available to the doctor to prescribe.
Is it true that 100% of those medicines are interacting with the patient chemically, rather than through protein folding?
So that if that’s the case, does that mean that where proteins misfold, we can’t combat it with any kind of folding algorithm, we just prescribe chemistry for your body to handle the impact of that.
Is that, did I say any of that right?
I think that is the majority of drugs, they are chemicals that we take, we are there, we are, you know, we take them as pills.
The chemicals are not going to fix the fold, it’s going to treat the symptoms of the misfolding that happened.
We’re not changing the mutations of the proteins, that could be something like gene therapy, but these are chemicals that come in and will attach themselves to these proteins and somehow mitigate the, you know, something like a misfold or it’ll change an interface, change how these molecular machines work.
Is there a particular disease that isomorphic labs are focusing upon right now, or is this more of a broad spectrum?
Let’s go for proteins and cherry pick out certain things, or will we be really looking at one particular?
The technology we’re creating is really, really general.
We want to be able to apply this drug design engine on any protein, any target, any disease area that comes our way.
Now, saying that, you know, practically as a company and you want to focus on a particular area, we’re focusing at the moment on, you know, a lot on cancer and a lot on immunology.
All right.
I’m too biggie.
And the two that probably lend themselves best to what you’re trying to accomplish, actually.
Okay, the question everyone’s going to want me to ask right now that’s watching this and listening to this is, how are you getting on?
I mean, it’s going really well, to be honest.
We’re seeing these algorithms actually, you know, change the way that we’re able to do drug design.
We’re able to discover, you know, completely novel chemical matter against some of these targets that people have been working on for even over a decade.
It’s really, really hard stuff, making amazing progress.
It’s still really early in the company, but it’s super exciting.
And have you sent anything to be photographed yet?
We sent some things for molecular photographs, yeah.
Okay, I know you’re not allowed to talk about it.
I know, I get it.
But we’re all very happy.
Okay, listen, I’m with you.
I’m picking up what you’re putting down.
That’s cool.
Yeah, but so if you, I see this work as fundamental research so that you publish a result, you publish the image as they published the image of the DNA molecule to know that it was a double helix.
Exactly.
That becomes public knowledge at that point.
So someone with tools, access to Alpha Fold 3, would any company have access to this once you have published the blueprint for it?
In drug design, often these blueprints come out in the patents.
So when you’re going to go into clinical trial, you need to patent these molecules.
And in those patents, you’ll have a lot of data around the molecules, the formula, how they work.
That’s what I was talking about earlier.
Yeah, yeah, yeah.
Okay.
Yeah.
All right.
So the immune system, the cancer, these are leading causes of maladies in this world.
What of the genetic disorders that affect one in 100,000 people?
Wow.
You bring them together, there’s enough of them, you know, they’ll fill a stadium in the world, but that’s so uncommon as to not really trigger anybody’s interest to sell.
It’s also not profitable.
Because you don’t have enough of a market there to sell the drug.
Right.
I mean, you know, it’s exactly that point, Chuck.
Traditionally, it might not be that attractive commercially to go after very small patient populations.
But in a world where it’s so much cheaper, so much easier to get to these drug molecules, then that opens up all of this space.
The cheaper it is, the easier you can justify going down that risk list.
This is a big guiding star for us.
This is why we’re doing this.
That’s beautiful.
I see what you did there, guiding star.
You know, it would be great though.
He’s picked up the environment.
He’s not silly.
Why is Chief AI Officer?
I’m interested in how active the company is in shaping policy around what you’re doing because there’s going to be a great deal of legislative policy that is going to be tied to what you’re doing.
All of the patent implications, there’s going to be research implications.
There’s going to be a lot of things tied to this.
We’ve been talking in this conversation about drug design, but then once you’ve designed the drug, you’ve got to go into patients in clinical trials, and that’s a really long process.
That’s why we have mice.
But even these mice models, they’re not actually very predictive.
You do all these studies in mice, and then it doesn’t translate into success in people.
You’ve got to go up the evolutionary scale and then get to the human bit.
Yeah, exactly.
And so you can imagine a world where we can design loads of new drugs.
We’ve got to be changing the way that we’re doing clinical trials, how we can actually get these drugs to patients who really, really need them in a timely manner.
So I think there’s a lot to be done and rethought there.
Is the ultimate goal for AlphaFold and I think medical science as a whole, to be able to bespoke medication for you as the individual rather than the broader spectrum medication that we find ourselves with all the side effects?
So are you able to then design a drug or a medication that has zero side effects and works exactly for me?
This is the goal, this is what we’re shooting for.
Imagine a world where we can sequence your particular cancer mutations and then based on those, your individual mutations, be generating specific drugs for you that even these are like 3D printed or something around the corner.
Okay, yeah, this is, I mean, we’re not there yet.
We’re in the very nascent stages of that right now with immunotherapy for cancer treatments and.
But how many of these yet to be cured diseases lend themselves to solutions that involve protein folding?
And how many are just plain old, old fashioned chemistry?
Proteins make up like pretty much all of our molecular machinery.
So there’s a class of disease which is due to misfolding, but then there’s many, many other diseases which are due to, for example, a protein not being expressed properly, or a cell going wrong in a certain tissue type.
If I have a bacterial infection, I give myself antibacterial chemicals, and then I’m done.
Do I need you for that?
But those chemicals are interacting with the proteins in the bacteria.
So proteins are the fundamental machinery, and the chemicals which are drugs are modulating those proteins, whether it’s like in our cells, in bacteria.
So basically everything you’re talking about is all happening on the cellular level.
If what you’re describing is happening inside of cells, proteins doing their thing, their 3D jigsaw puzzle, and you have a solution for that, a remedy, you have to get your remedy inside the cell to interact with that folding.
The delivery system.
And how do you do that other than through like a Trojan horse virus or something?
Because viruses get in there pretty on command.
Yeah, well, if you think about the drugs that you take as pills, drug design is really hard because it’s not just about targeting these proteins, we’ve got to get them to the right place.
We might want a pill that you take.
So you take this pill, it’s got to be absorbed by the body.
So it’s got to be soluble, it’s got to go through the gut wall, it’s got to go through the bloodstream to the particular tissue type, the cell type you care about.
Then it’s got to go through the cell membrane to be able to actually target maybe a target which is within the cell.
So you need all of these properties in a single molecule.
So we’re actually designing these molecules not just to hit the protein, but also to be soluble, to be cell permeable.
There’s so many different factors.
And then you don’t want this molecule to be toxic.
So you want it to hit the target of interest, but not hit anything else.
Right.
So I can see how a molecule gets through the cell wall.
A simple molecule, but a full up protein, red-blooded protein, how’s that getting through the cell wall?
Exactly, there’s different types of drugs.
Some are what we call small molecules, things you could take as pills.
Others are made from proteins.
There, they’re often things that you would inject directly inside.
And some of those might be cell permeable.
They would be things like peptides.
But often these protein-based drugs, things like antibodies, they’re injected, but they don’t go in the cell.
They’re just interacting with proteins on the surface of cells.
So you don’t need that permeability.
So it really depends on what your target is.
And how do you want people to be able to take that drug?
Sometimes a pill is the best thing, but actually sometimes injecting is the best thing.
And remind me what a peptide is.
A peptide is a really small protein.
So you’ve got full-blown proteins, which are these big molecular machines, and then you’ve got small proteins made up of five to 50 amino acids.
Those are peptides?
Those are peptides.
They’re smaller, so sometimes in some configurations they can get through the cell wall.
So, with this computer science, are we upending chemistry as we’ve known it?
And are we going to find it kind of moving off into…
With AI?
Yeah, with the AI, is it then sort of moving into the other sciences?
Are we going to just see it stick in one particular area?
Chemistry is always going to exist.
It’s like, to me, it’s like any field of science at the moment.
Doing science like chemistry without math, you wouldn’t think about that now.
And it’s going to be the same thing with AI.
It already is, in my mind.
You just wouldn’t do chemistry without AI.
You wouldn’t do biology without AI.
It’s just that fundamental tool that allows us to understand the world better.
So chemists will not one day be like coal miners.
Just like, I remember my grandpapa used to go into the lab.
So going back to an issue Chuck was mentioning.
He come home smelling like chemicals.
Who’s actually going to be able to access AlphaFold 3 or AIs of this iteration?
Is it exclusively isomorphic labs or this comes out in license?
I want a home kit.
That’s it, exactly.
My DNA goes in, there’s an AlphaFold going on, out comes a pill and I take it and I don’t even need you at that point.
That would be so cool.
You just finger prick or put your finger on a sensor or something and it figures it all out.
It makes your own pill at home.
Yeah, look up the Theranos story for that one.
Wow, that’s the woman that went to prison, right?
But no, you can access AlphaFold.
So if you search for AlphaFold server, there’s a whole web-based system where you can fold proteins there for academic uses.
It’s really cool.
So you can just put your system in, get the 3D fold out from AlphaFold 3, download it.
Oh, cool, man.
I mean, how far away are we from modeling an entire human being, which I suppose touches on to your fears?
Modeling or creating?
Either or.
Once you model, the next step is creating.
That’s true.
That’s all there is to it.
That’s true.
That’s the dream.
We kind of need to work up the scales here.
So we can model how two atoms interact.
We can write down those equations.
We can simulate small atomic systems.
With things like AlphaFold, we get into bigger atomic systems, things on the scale of multiple proteins.
Now we’ve got very accurate AlphaFold 3.
Maybe we can actually bootstrap off that to get to more, even bigger systems, what we call pathways, how all of these things interact.
It’s only just now clicking within me because you can look up in a book, the tables, of action potentials for the interactions of atoms and molecules.
And so you’d have a very good sense of which molecules will combine.
Is it exothermic?
Is it endothermic?
But these are atoms and molecules.
And as powerful and as convenient as that is, that’s just the first rung in this ascending ladder of complexity that you are gaining control over.
Yeah, and there’s trillions of atoms within a single cell, let alone the whole human body.
It’s just unfeasible to simulate the whole thing.
But what you can do is you can, we do have good measurement techniques at different levels of scale, so we can measure things like protein folding.
We can measure the amount of protein within a cell.
We can measure the number of cells of certain type within a tissue.
And so we have these like-
The bigger it is, the easier it would be to measure.
So we have these little windows into this sort of microscopic world, and then we can use AI to sort of fill in the gaps and bootstrap off the stuff we can do well, the atomic level, and start building up that scale of modeling, if that makes sense.
We can rebuild them.
I read this article like, God knows how long ago.
God, how long did you read the article?
God, because you know God.
Anyway, it was talking about when a fertilized cell starts to proliferate and become a person.
And basically, what it determined, what the scientists determined at that point, and this is many years ago, is that the only way they could describe it is, there’s a bunch of noise.
Like, there’s just a bunch of noise.
We can’t really see anything.
We can’t make sense of any of it, because it’s just basically, if we were to look at it as data, it would just be noise.
Are you able to pierce that veil and see into that?
I mean, we haven’t been looking at that specific thing.
Okay.
But this is where you start to understand more about, you know, a really granular scale, and then you can integrate that and create these sort of, I don’t know, coarser measurements and coarser predictions.
This is what we do in lots of areas of science, right?
We don’t simulate the whole universe at the atomic scale, but we find these rules of thumb or ways to describe sort of broader collections of molecules.
And that’s what we can start to build up and actually learn with these neural networks.
Cool.
So, question on behalf of Chuck.
Could AlphaFold discover a hallucinogenic that could make him see God?
Or any other deity or being?
Thank you for asking.
Welcome.
I would like a very real answer, please.
You can go on the AlphaFold server and try that out.
Okay.
All right.
Invitation.
Hey, listen, I’m all about it.
So we’ve looked in previous shows, talking to Biomedical Engineering, and if we are to travel off world and deep space, we are probably going to need different upgrades for us to be able to do that.
Are we going to be able to, with AlphaFold or AI like this, be able to upgrade ourselves to make this sort of deep space travel?
Or upgrade ourselves for anything?
Any need that we’re otherwise…
Yeah, I mean, this is a sci-fi, you know…
Yeah, now we’re sort of beyond, you know, solving disease into like, actually, can we enhance ourselves?
Right.
I don’t know, I think like, there’s probably potential, right, to think about creating chemical matter that we can take or ingest.
I mean, aging alone would be a huge application for this.
I mean, there’s crazy research on aging.
Aging is basically cellular degeneration, and if you’re able to, on a molecular level, kind of restart that process or jump-start it or boost it.
Aging is an interesting one, you know.
This is a really nascent area of research where people are just starting to work out what are some of the factors that reverse the age of cells.
There are these things called Yamanaka factors.
And there’s even potential that people are finding of creating molecules that stabilize particular protein.
Yamanaka factors are proteins, they’re transcription factors that read DNA, can stabilize these things.
Maybe that is what reverses some of the age of cells.
This is super nascent.
So what is the connection between wanting to modify a genome and your ability to fold proteins to interact with our physiology?
I ask that because I’m reminded there was a scene in the film Gattaca where they didn’t manipulate your genome but they selected your pre-existing genome for certain properties.
And there’s a person giving a piano recital and a very rich sound.
I mean, it was beautiful.
And then the camera came around to the front and the person had 12 fingers.
That’s right.
And bred for that.
Yes.
Right, you get two extra notes for every For everything.
For everything going on.
She could only play the stuff, like nobody could play what she could play.
Nobody could play what she could play.
So, this would be modifying not to go into space necessarily, but just to sort of enrich the diversity of the human species.
We’re not doing genetic modification.
So he says.
He’s English.
You must trust him.
To be honest.
Thank you.
Well done.
Well played.
Can you?
Is it the same thing?
Some particular types of drugs are things that would manipulate your genome.
That’s how people start to target some diseases.
This is not the class of drugs we’re working on.
When we think about the big ambition of solving all disease, maybe this is something that we need to be doing over time as we want to really crack the whole spectrum of disease.
Is it even possible to consider that without considering the whole area itself as it all bleeds in together at some point?
Yeah.
I mean, we need to understand the genome and all the effect, how changing a particular base pair on your DNA is going to change what proteins are expressed or in what abundance and how all the knock-on effect on the pathway is.
You really want this, basically, this virtual cell to be able to manipulate this cell on a computer to do experiments there.
Here’s what I want you to do.
It’s like your cell template that you’re talking about.
I’m talking about prioritizing.
And I’m not asking much because it already happens in the animal kingdom.
You know, for so long, decades, even centuries, we imagined ourselves at the top of some evolutionary construct.
With the apex of the triangle.
Without any arrogance whatsoever.
And, all right.
Yet, a newt can regenerate a limb and we can’t.
And so it seems to, and they’re vertebrates.
So it seems to me there ought to be some way to extract from animals that do things that we could benefit from and then make that a priority.
So people, especially veterans who’ve lost limbs in conflict.
Or even geckos with their sticky hands.
Like maybe I could be Spider-Man one day.
So all going to end up as-
Let’s prioritize that, Chuck.
We’ll become superheroes.
So the regeneration of limbs, that’s got to be a protein thing going on in there, isn’t it?
Yeah, I mean, all of our mechanisms are proteins.
Same for newts as well.
So there is some mechanism there.
I don’t know what it is.
Okay.
But that would be a mechanism to emulate, if you could.
If you could, yeah.
And then install it into our own physiology.
Big if.
Yeah, that’s a lot.
How about this?
Would you be able to look at drugs that are already here?
And there are some drugs that are just not well tolerated.
And you’d be able to reconfigure them in such a way that you get the benefit of the drug without the side effects.
Yeah, exactly.
So you often have these first generation of drugs that do something, but they have these side effects.
Then there’s a big opportunity to understand better how these drugs work.
Things like Afrofob-3, things like our models that understand toxicity of drugs can then allow us to potentially modify these to become better drugs and have less side effects, less toxic effects.
Cool.
Well, that’s going back to the medical catalogue and reanalyze it, which is exactly what an AI would be perfect for.
Right, exactly.
Yeah, you guys are going to make a lot of money, man.
I don’t know how I get…
Stop seeing dollar signs and see some…
How do I get a piece of this company?
I need to get a piece of this company.
You guys are going to make…
I mean, I can’t even imagine the amount, the gobs and gobs of money.
More money than cells in my body.
This is amazing.
One of the doorstep of quantum computing, and I know what impact that would have in my field.
In your field, would it make your entire life’s work look like it was done on an abacus?
Yeah, I mean, this is going to change things.
I think open question how this changes machine learning.
What can AI do with quantum computing?
But for chemistry, even near-term, there are some real applications of quantum computers for understanding the properties of small molecule drugs.
Because actually, some of the things that people do today with quantum computers is simulate these small chemical systems.
We actually, even in the company, we have a quantum simulation team that are not using quantum computers, but simulating the quantum effects of molecules.
Now, if you had a quantum computer that could work on that scale, you could use that instead.
Wow.
I think of so many needs on the frontier of chemistry in modern society.
One of them is, what do we do with all the plastic that’s in our environment that’s still there in the ocean?
Is there some life form you can create that will digest the plastic and turn it back into its original molecules?
Are proteins something that could be applied there?
If not in your world, then you’re describing an ability more than you’re describing a specific solution to a problem.
You’re empowering the chemist in ways never previously imagined.
Yes.
So you can use the capability of alpha fold to understand the structure of proteins.
People are using this outside of drug design.
People are using this, for example, to create bacteria that have enzymes that could potentially digest plastics, like you’re talking about.
You could think about this for engineering more resilient types of crops, these sort of things.
So just like AI, this is a platform upon which you can rest the technology of any field.
That’s the amazing thing about the protein folding problem.
Once you start to solve that, you unlock so many new things for a whole broad spectrum of science.
There’s a lot of downstream benefits, a good shame.
Okay, last thing.
Here’s the last thing.
How do I get a piece of this company?
No, Chuck is good.
Dollar times.
How do I get a piece of this company?
Last thing.
Last thing.
What is the worst possible outcome of your work?
Ooh, what a question.
What guardrails are necessary as we go forward?
Because any new technology with awesome power comes awesome responsibility.
Yeah, I think you have this with AI, you have this, you know, creating new biology or chemicals.
You just need to think about how to use this responsibly, like what you’re putting out into the world openly versus what you close off for many safety reasons.
I think there’s a lot of things to consider there.
Because famously in one of the Jurassic Park films, they withheld lysine, amino acid, from one of the dinosaurs in case it escaped.
It would die because it would need the lysine for its survival.
And that was a kind of an insurance plan that put in, but life always finds a way.
Yeah.
There you go.
Anyway, Maxwell Jaderberg.
Yeah.
Thank you for joining us on StarTalk.
We’re going to be watching your company and Chuck wants a piece of it.
Yes.
I don’t know what that means, but anyhow, this is, I’m delighted to just be able to look through your lens at the birth of an entire frontier in human physiology.
I mean, what a time this is.
No, thank you so much.
It’s been super fun to talk.
No, thank you.
Excellent.
Wonderful.
All right.
I think we’re done here.
It’s been another installment of StarTalk Special Edition.
Talking about AI, human physiology and the future of trucks.
Oh yeah.
Gary, good to have you always.
Pleasure, Neil.
Chuck, always a pleasure.
As always, this has been StarTalk Neil deGrasse Tyson, your personal astrophysicist.
Keep watching.

 Unlock with Patreon
Unlock with Patreon



 Become a Patron
Become a Patron